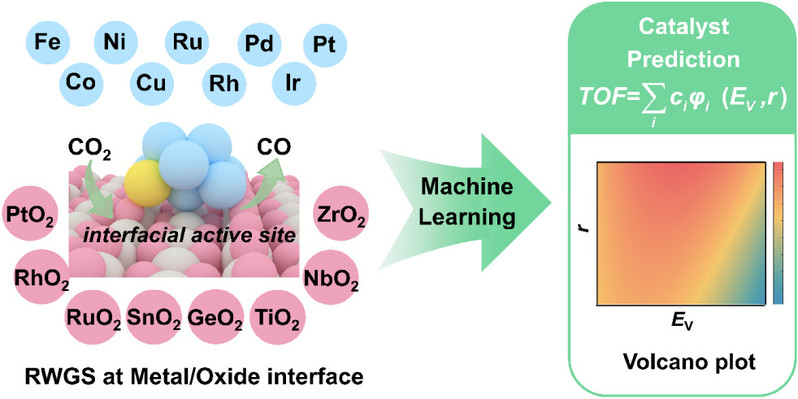
The dynamic behavior of metal-oxide interfaces under reverse water-gas shift (RWGS) conditions is crucial for rational catalyst design but remains poorly understood. Through first-principles calculations and interpretable machine learning, we reveal how reactant-induced stabilization governs structure-activity relationships at these interfaces. Our study identifies two key descriptors—oxygen vacancy formation energy (EV) of the oxide support and atomic radius (r) of the supported metal—that collectively determine RWGS performance. Activity exhibits a volcano-type relationship with EV, peaking at ~3.4 eV. The reaction mechanism shifts from carboxylate-mediated pathways on low EV oxides to direct CO2 dissociation on high EV supports. Larger metal atoms (Pt, Pd, Ru) stabilize transition states by shortening O-H bond distances, significantly lowering barriers for lattice oxygen hydrogenation. Oxygen vacancies play dual roles: stabilizing intermediates and promoting vacancy regeneration throughout the catalytic cycle.
These findings establish a physically-grounded predictive framework linking reactant-induced interface dynamics to catalytic performance, enabling rational design of efficient RWGS catalysts.